When a chemical species contains an unpaired electron, electron paramagnetic resonance(EPR) or electron spin resonance(ESR) spectroscopy is the technique that is used to study unpaired electron compounds.
Principal:
The phenomenon known as EPR is based on the fact that an electron is a charged particle that has the ability to spin around it’s axis causing it to act similar to a tiny bar magnet.
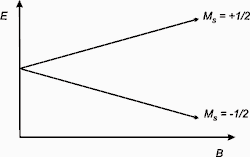
when a strong magnetic field(Bo) is applied to a molecule or compound that contains an unpaired electron, the spin of the unpaired electron can align in two different ways creating two spin states ms= + 1/2, -1/2. This energy difference is due to the Zeeman effect.
The alignment can be either along the direction(parallel) to the magnetic field corresponding to the lower energy state m= -1/2 or it can be opposite(antiparallel) to the direction of the magnetic field m= +1/2.
These both alignments have different energies, and this difference in energy lifts the degeneracy of the electron spin states.